Masipag, 19 December 2019 | Source
In a sly move characteristic of the GM proponents including the various agencies in the Joint Department Circular (JDC), the direct use for food, feed and processing of the genetically modified (GM) Golden Rice has been approved on December 10 despite the staunch opposition from farmers and consumers in the Philippines. MASIPAG condemns the Golden Rice approval and the collusion among the International Rice Research Institute (IRRI), Philippine Rice Research Institute (Philrice) and the Dept. of Agriculture-Bureau of Plant Industry (DA-BPI) which gravely threatens not only the health of the people and the environment, but also the future of rice production and farmers’ control of seeds.
Farmers and consumers in the Philippines and in other countries as well, remain adamant that Golden Rice will not address the Vitamin A Deficiency among vulnerable sectors in developing countries, but is in fact a tool of the increasing corporate control over agriculture. By harking on the humanitarian packaging of the Golden Rice, and marketing it as ‘Healthier Rice,’ proponents are deceiving and blinding the people from the ulterior intention of Golden Rice which is to usher in more genetically modified food such as soybean, cotton, potato and more varieties of GM corn. According to IRRI’s website, research is also being conducted on high-iron and zinc rice in response to iron-deficiency anemia and stunting.
“We are appalled that the approval has pushed through despite the numerous issues that MASIPAG and many other organizations are raising regarding Golden Rice, mainly on its safety and long-term effects on children, women and other consumers,” said Elpidio Paglumotan, a rice farmer from Negros and Chairman of the Board of Trustees of MASIPAG. MASIPAG, along with other farmers’ and consumers’ organizations has previously written to DA-BPI on October 16 to discuss the consolidated risk assessment report and the comments from respected scientists including Dr. Chito Medina, Dr. David Schubert and Dr. Michael Antoniou contesting the proponents’ claim on the safety of Golden Rice.
“Up to now, proponents have failed to address concerns on the Golden Rice’s negligible beta-carotene content, its fast degradation and the possible toxicity associated with the beta-carotene degradation,” said Cris Panerio, National Coordinator of MASIPAG. “Instead, they hide behind the supposed ‘rigorous biosafety assessments’ and the debatable safety clearances from the US, Canada, Australia and New Zealand.”
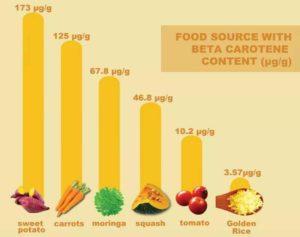
Golden Rice’s paltry beta-carotene content pales in comparison to local foods.

The meager beta-carotene in Golden Rice degrades fast after processing and long storage time.
The lack of transparency and speed of approval of Golden Rice reeks of the desperation of IRRI, Philrice and DA-BPI to bring in the GM rice which has been gaining greater opposition because of it is unnecessary and unwanted. “Such acts are in dissonance to the spirit of the JDC on transparency and public consultation,” added Panerio. MASIPAG calls the attention of the newly-appointed DA Secretary William Dar, as well as the other members of the JDC to put the welfare of the Filipino people instead of the corporate-influenced research institutions.
Also read:
